Investigation of stability of colorant powders extracting from Japanese purple sweet potato (Ipomoea batatas (L.) Poir)
- Ho Chi Minh City University of Technology, VNU-HCM
Abstract
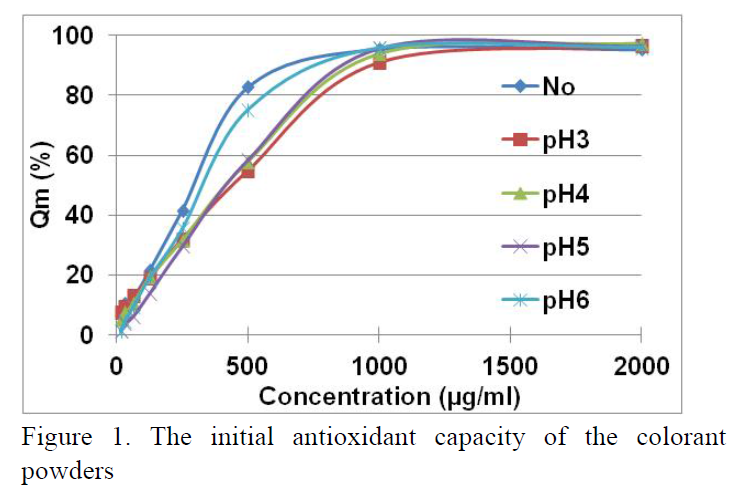
The stability of colorant powders from Japanese purple sweet potato (Ipomoea batatas (L.) Poir) harvested in Vinh Long province was investigated. The colorants were formed from extracting solutions which were adjusted to pH of 6, 5, 4 and 3 before converted to powder matrix. They had red-purple color and contained anthocyanin concentration of about 32 mg/g. The IC50 inhibitory concentration of the neutral powder was 292.7 μg/mL, whereas the IC50 of pH3 powder was 484 μg/mL, which antioxidant activity decreased by increasing of acidic media clearly. Stability of appearance colors, anthocyanin content, polyanthocyanin index and antioxidant capacity were investigated and they depended on storing conditions (temperature and radiation) and pH media. To avoid denaturation of anthocyanin content, the pigment should be stored in the dark and at low temperature (10 °C). The acidic media could preserve the colorant powder by protecting anthocyanins, but it reduced the biological property of the pigment.

