
Studying the influence of solvent concentration on the fabrication of PCL membrane for wound dressing applications
- School of Biomedical Engineering, International University, Vietnam National University Ho Chi Minh city
Abstract
Introduction: The aim of this study is to evaluate the influence of the concentration of acetone (AC) and acetic acid (AA) solvents to fabricate a polycaprolactone (PCL) membrane for wound dressing applications. The viscosity of PCL solutions is evaluated to choose the best AC and AA solvent ratio.
Methods: The PCL membrane was successfully fabricated by the electrospinning method. The physicochemical properties of the membranes were evaluated using a scanning electron microscope (SEM), fiber diameter distribution, water contact angle measurements, the moisture vapor transmission rate, and a biocompatibility assay.
Results: The results show that two samples of 15%PCL-AC and 12%PCL-AC are suitable for fabricating the membrane by electrospinning method.
Conclusion: Therefore, two samples consisting of 12%PCL-AC and 15%PCL-AC with good physical-chemical properties and good biocompatibility can be selected for use in wound dressing applications.
Introduction
Skin is an extremely important organ that protects the human body. While it is often thought of as just an exterior cover, there are various important reactions that take place on the human skin such as microbial defenses, hydration, and thermal control. The innate immune system's first line of protection against infection is the skin, which reduces the likelihood of microbial attacks1, 2. However, skin is easily prone to external injuries such as bruises, burns, cuts, lacerations, gashes, and tears, causing wounds. When the skin is wounded, one of the major complications is infection, which makes the wound progress from an acute to a chronic condition if the wound site is not treated as soon as possible 3. Choosing a suitable wound dressing is necessary and important. Various types of wound dressing have been researched by scientists and are manufactured using many different methods in which electrospinning has been applied for gauze fabrication in recent decades4. This method is very convenient and has the ability to be used for a variety of polymers with different levels of biological compatibility such as cellulose, gelatin, silk, polyvinyl alcohol, polycaprolactone (PCL), etc5. In addition to being able to be used in traditional dressings, the electrospun membrane has demonstrated advantages such as breathability, easy synthesis, and especially the easy addition of other substances to increase the antibacterial abilities and wound healing support. Among the types of materials used for wound dressing, PCL membranes are outstanding thanks to their mechanical properties, high biological compatibility, and electrospun PCL membranes that hardly deform when influenced by body temperature and room temperature6. Electrospinning technology is a simple technology with the ability of high economic efficiency because of the ability to create thin membranes. This saves on the cost of materials during production. Moreover, this technology is gradually becoming popular in the medical industry due to its versatility with many materials. From this process, we can produce different healthcare products using electrospinning which can match the required international quality at lower prices. Electrospinning uses an electrospinner in which the machine structure includes (1) a one-way high-voltage power supply, (2) the fiber collection system, and (3) the electrical syringe system containing the polymer. Electrospinning can produce nanofibers by applying high electric fields. When a high enough voltage is placed in a drop of liquid, the liquid will be charged, and the electrostatic force thrusts against the surface tension from which the liquid drops are stretched to form a spray connected to the membrane collector. Overall, electrospinning is a set of electric fields that control the polymer spray rays in relation to the membrane collection block that forms a non-woven format product. Electrospinning products obtained with a microstructure consist of countless interwoven polymer fibers with a diameter of several hundred nano to several micrometers. The parameters include the properties of the polymer solution (molecular weight, electrical conductivity, conformation of polymer chains, surface tension, and solvent type) while the process parameters include applied voltage, the distance between the needle and collector, electrode geometry, the rotating speed of the collector, the feeding rate of the polymer solution, and the environmental conditions (temperature and humidity)7. The viscosity of solution is one of the most important properties8. For a successful electrospinning procedure, the selection of an appropriate solution viscosity is among the key parameters because continuous fibers cannot be produced with a solution consisting of low viscosity. At a high viscosity, the electrical charges fail to generate the required strength to attenuate the solution to form fibers. With an increase in viscosity, the amount of polymer chain entanglement of the solution also increased and the shape of the beads transformed from spherical to spindle- like until a smooth fiber was produced. It has been found that the optimum spinning viscosity ranges from 1 to 200 poise approximately a range of 1 to 20 uniform nanofibers can be produced9. Therefore, the aim of this study is to evaluate the influence of solvent concentration on the process of creating PCL membranes using the electrospinning method in relation to wound dressing applications.
Materials and methods
Materials
Polycaprolactone (Mn= 80000, Sigma-Aldrich), Acetone (>99,5%, Xilong), acetic acid (>99.9%, xilong), acetonitrile (>99,9%, Sigma-Aldrich), Dulbecco's Modified Eagle Medium (Thermo Fisher), Fetal Bovine Serum (Thermo Fisher), Penicillin-Streptomycin (Thermo Fisher), and Resazurin (Thermo Fisher) were used.
Methods
The PCL membrane was successfully fabricated using the electrospinning method. The physicochemical properties of the membranes were evaluated using a scanning electron microscope (SEM), fiber diameter distribution, water contact angle measurements, moisture vapor transmission rate, membrane surface hydrophilicity, and a biocompatibility assay.
Scanning electron microscopy (SEM)
The membranes (1x1 cm) were sputter coated with gold (JEOL Smart Coater, Japan) for 60 seconds. The SEM (JSM-IT100, JEOL, Japan) with an acceleration voltage of 10kV was then used to evaluate the surface and side morphologies of the membranes. The fiber diameter, pore size, and thickness of the membranes were analyzed using the ImageJ software (NIH, USA).
Water absorbability
The water contact angle measurements were taken according to the following protocol. Principally, 10 μl of water droplets were dispensed onto the surface of the membranes using a pipette. Immediately, any change in water droplet shape during the testing period was recorded using the contact angle set including the DSLR camera (Canon) together with a pedestal system. The contact angle of the recorded images was analyzed using the ImageJ software (NIH, USA). The experiment was performed in triplicate at three different locations on the surface membranes.
Moisture Vapor Transmission Rate (MVTR)
The Moisture Vapor Transmission Rate (MVTR) of the membranes was carried out according to BS EN 13726-2:2002. A cup filled with distilled water was completely covered by the testing membrane of 6x6 cm (A), provided that the membrane was placed 5mm above water level. The system, including the container, membrane, and water, was then weighed (W1, g) before being incubated in an oven at 37C for 24 hours (T). After that, the whole system was weighed again (W2, g) and the dressing was observed. Each test was replicated five times. The Water Vapor Transmission Rate (MVTR) of the samples was calculated as follows:
Tensile strength test
The mechanical strength of the membranes was tested using Texture Analyser (TA.XTplus, Stable Micro Systems, USA). Firstly, specimens with a rectangular shape of 1x3 cm were prepared from the membranes. Prior to testing, the thickness of the membrane was measured using an electronic caliper. The stress and strain graphs of the membrane were reported. The measurements were repeated three times and the data presented as average ± standard deviation.
Biocompatibility assay
First, the membranes (1 cm x 1 cm) were soaked in Dulbecco's Modified Eagle Medium (DMEM) cell culture medium (ratio of 0.1g/mL) with 10% bovine serum (Fetal Bovine Serum, FBS) and 1% antibiotic (Penicillin, PS) for 24 h to collect the extracted solution. Secondly, the extracted solution was used to culture L929 mouse fibroblasts in a 96-well culture plate at a concentration of 10 cells/100μL/well for 24 h. After that, resazurin was added to each well at a concentration of 2 μg/100μL and incubated for another 4 hours. Finally, the fluorescence signal was measured at an excitation wavelength of 530 nm and absorption wavelength of 590 nm. The experiments were repeated three times.
Statistical analysis
The statistical analysis was performed using Sigma Plot V.12.0 version (SSI, USA). Differences between samples were analyzed using one-way analysis of variance (ANOVA). The data was expressed as the mean ± SD and P values, while those that were less than 0.05 (p ˂ 0.05) were determined to be statistically significant.
Table of viscosity ratings of PCL solutions at various concentrations of 12%, 15% and 20%, dissolved in AA, AC solvents and mixed solvent systems of 7AA:3AC, 8AA:2AC and 9AA:1AC (n=10)
|
12% PCL |
15% PCL |
20% PCL | |
|
AC |
225 ± 4.96 |
450 ± 4.96 |
1940 ± 31.04 |
|
7AA:3AC |
400 ± 9.04 |
700 ± 6.64 |
3190 ± 35.84 |
|
8AA:2AC |
445 ± 11.04 |
765 ± 18.56 |
3270 ± 20.16 |
|
9AA:1AC |
470 ± 17.84 |
1050 ± 16.56 |
3475 ± 26.4 |
|
AA |
515 ± 9.84 |
1625 ± 9.36 |
3500 ± 18.32 |
Results
More specifically, increasing the AA ratio in the series of solvent mixtures yielded greater viscosity ratings for the newly synthesized 12% PCL membrane (from 225 cP with no AA present up to 515 cP with only AA present in the solvent). Moreover, in the previous work of the group10, PCL membranes were successfully using AC solvent at a 15% PCL concentration corresponding to 450 cP viscosity. In this study, 450 ± 4.96 cP was chosen as the control value and the solution samples with an approximate viscosity value (12% PCL-8AA:2AC) were investigated using the new electrospinning system. A solution of 12% PCL-7AA:3AC, 12% PCL-9AA:1AC and 12% PCL-AA with viscosity values of 400 ± 9.04, 470 ± 17.84 and 515 ± 9.84 cP respectively were also chosen due to their resistance regarding the clogging of the nozzle (no need to clear the needle)11. In addition, a solution of 12% PCL-AC with a viscosity value of 225 ± 4.96 cP, was also selected for its good membrane forming ability from the experiment. Therefore, the six solution samples of 15% PCL-AC, 12% PCL-AC, 12% PCL-AA, 12% PCL-9AA:1AC, 12% PCL-8AA:2AC and 12% PCL-7AA:3AC were chosen and the other remaining samples were not selected.
Table of records from membrane synthesis and membrane bombardment efficiency ratings during the assessment procedure of the effects of PCL concentration and its solvents
|
Voltage and Observation Distance |
Cleaning of Injector Head |
Bead formation |
Efficiency Rating (%) |
|
15%PCL-AC |
+ |
- |
89 ± 1.04 |
|
12%PCL-AC |
+ |
- |
90 ± 1.40 |
|
12%PCL-AA |
- |
+ |
83 ± 1.44 |
|
12%PCL-9AA:1AC |
- |
+ |
84 ± 1,36 |
|
12%PCL-8AA/2AC |
- |
+ |
89 ± 1.04 |
|
12%PCL-7AA/3AC |
- |
+ |
97 ± 1.04 |

Overview of the effects of PCL concentration and solvent composition on the morphology of membrane surface (a) 15%PCL-AC, (b) 12%PCL-AC, (c) 12%PCL-8AA/2AC, (d) 12%PCL-7AA/3AC, (e) 12%PCL-9AA:1AC and (f)12%PCL-AA (Measuring scale: 10 µm))

Distribution diagram of samples’ strand and pore sizes (a) PCL12%-AC và (b) PCL15%-AC (n=30)

Cross section scans and distribution diagrams of membrane thickness of samples (a) 12%PCL-AC and (b) 15%PCL-AC (Measuring ruler: 100 µm, n=30)

(a) The hydrophobicity, (b) the mechanical, and (c) the biocompatible properties of membranes
The average sizes of the membranes synthesized from 12% PCL-AC and 15% PCL-AC were 0.88 ± 0.40 µm and 2.29 ± 0.34 µm. On top of that, the average pore sizes were 2.13 ± 0.64 µm and 8.66 ± 2.69 µm.Figure 2 shows that when the 12% PCL solution in AC was used, 33,33% of all PCL strands had diameters of 0.4 - 0.6 μm and nearly 50% of the pores spanned 1.5-2.0 μm across in diameter. Nevertheless, as the concentration of PCL in the solution increased from 12% to 15%, the diameter of the strands, in conjunction with the pore sizes, were observed to increase in lock-step. Additionally, strands with diameters of 2.2-2.4 μm made up 36.67% and pores with diameters of 6-8 μm made up nearly 50%.
SEM scans and distribution diagrams of the membrane’s thickness for the two samples 12% PCL-AC and 15% PCL-AC are illustrated in Figure 3. The results were indicative of a uniform thickness across the membranes synthesized by the electrospinning device with negligible inconsistencies between the measurements. Under the same injection conditions and solution volume (45mL), the membrane made from 12% PCL-AC averaged around 230.7 ± 20.1 µm of thickness less than the membrane fabricated from the solution 15% PCL-AC of 301.3 ± 19.3 µm (p < 0.05). This was mainly due to the average strand size of the membrane from 12% PCL-AC being much less, leading to an overall reduction in thickness.
Table of average evaporation rates of the samples 12%PCL-AC và 15%PCL-AC (n=5)
|
Sample |
Average evaporation rate (g/m2.24h) |
|
12%PCL-AC |
3690 ± 283 |
|
15%PCL-AC |
3866 ± 141 |
Figure 4a shows that the 12% PCL – AC and 15% PCL - AC membranes acquired hydrophobicity (with contact angles greater than 90). More specifically, the less hydrophobic surface was found for the 12% PCL membrane synthesized in AC solvent with a contact angle of 116.7 ± 5.6. The 15% PCL - AC membrane was more hydrophobic, leading to a widened contact angle of 127.1 ± 5.4. Besides, Figure 4b shows that the PCL 12%-AC membrane possessed a tensile strength of 4.011 ± 0.380 MPa and a strain of 672,900 ± 44,028%, both of which are significantly greater than some of the physico-mechanical properties of the PCL 15%-AC membrane with a 2.594 ± 0.250 MPa tensile strength and strain of 579,491 ± 97,284%. Finally, Figure 4c illustrates that both samples of 12% PCL-AC and 15% PCL-AC obtained good biocompatibility with the L929 cell viability maintained above 90% at a 100% extract concentration. The results showed statistically significant differences in cell viability across the three different concentrations of 100%, 50%, and the control in the 15% PCL-AC sample (p < 0,05). Overall, cell viability remained over 90% across all samples. The results indicate that the samples including 12% PCL-AC and 15% PCL-AC are biocompatible.
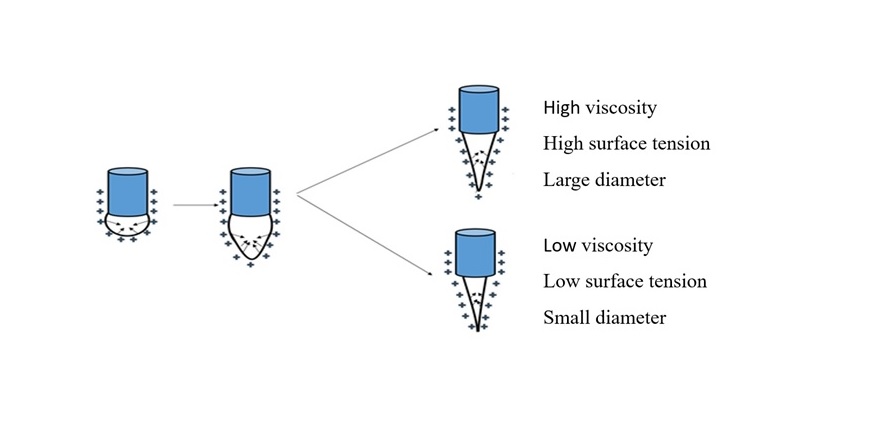
The effect of viscosity on fiber size.
Discussion
The concentration, viscosity, and surface tension of the solution are the factors that determine the morphology of the fibers. Surface tension tends to reduce the surface area of the droplet and the viscosity helps the droplet resist deformation when subjected to external influences. In contrast, electrospinning can increase the surface area of the droplet via droplet elongation (by the Coulomb force) to form polymer fibers. Therefore, the higher the surface tension and the higher the viscosity, the more difficult it is to stretch the droplet to produce fibers with a larger diameter (Figure 5)12. This could account for the increase in PCL concentration from 12% to 15% with the increase in surface tension and viscosity leading to an increase in fiber size.
Viscosity has been listed as one of the more impactful factors that mainly impinges on the formation of polymer strands and other characteristics such as the diameter and formation of polymeric beads on the strands. In addition, it was agreed that the greater the solvent’s viscosity, the higher the average size of the polymeric strands. In our previous study, a PCL membrane was successfully synthesized using PCL in AC which yielded a viscosity rating of 450 cP 10, 11 ; samples with a suitable viscosity were chosen. When varying the concentration of PCL in solution across the different samples including 15% PCL-AC, 12% PCL-AC, 12% PCL-8AA/2AC, 12% PCL-7AA/3AC, 12% PCL-9AA:1AC, and 12%PCL-AA, there was an increase in fiber diameter as well as pore size. The number of beads increased significantly as the different AA concentrations were increased (Figure 3). Besides, the 2 membranes of 12% PCL-AC and 15% PCL-AC possessed great hydrophobicity due to their angles of contact being greater than the offset of 90. A possible explanation can be because the pore sizes of the 15% PCL-AC membrane were more prominent (Figure 4a), prompting an inflation in hydrophobicity as more water droplets were pushed to the surface, resulting in much larger contact angles. In addition, the two membranes of 12% PCL-AC and 15% PCL-AC met all the preset physical conditions at a relatively satisfactory level where the ultimate tensile strength must be no less than 1.8 MPa with a deformation capacity of more than 30%. However, the 12% PCL-AC membrane was thinner, retaining greater tensile strength and elasticity (Figure 4b). Therefore, the 12% PCL-AC samples were chosen for use in wound dressing applications.
Conclusion
Two samples of 15% PCL-AC and 12% PCL-AC were determined to be suitable for fabricating membranes and both samples exhibit prominent biocompatibility where the cell viability is over 70%, which is needed to apply it to a wound dressing. However, the 12% PCL-AC sample is a more suitable option for use in wound dressing applications than 15% PCL-AC and the other samples evaluated in this study.
Competing interests
The authors declare that they have no competing interests.
Acknowledgement
This research was funded by the Department of Science and Technology of Ho Chi Minh City under the grant number 01/2020/HĐ-QPTKHCN. This work was also supported by Vietnam National University under the grant number NCM2020-28-01.
Authors’ contribution
Thi-Hiep Nguyen: Conceptualization, project administration; Van Khiem Nguyen: Data curation, formal analysis, visualization; Nghi Nguyen: Writing – original draft, Tam Phan: Writing – original draft, data curation; Phuc H.Vo: Writing – review and editing, formal Analysis; Thao-Nhi Dang Ngoc: Writing – review and editing; Kieu Nguyen : formal Analysis. All authors discussed the results and contributed to the final manuscript.
Abbreviations
AA: Acetic acid
AC: Acetone
DMEM: Dulbecco's Modified Eagle Medium
FBS: Fetal Bovine Serum
MVTR: Moisture Vapor Transmission Rate
PCL: Polycaprolactone
SEM: Scanning electron microscopy

